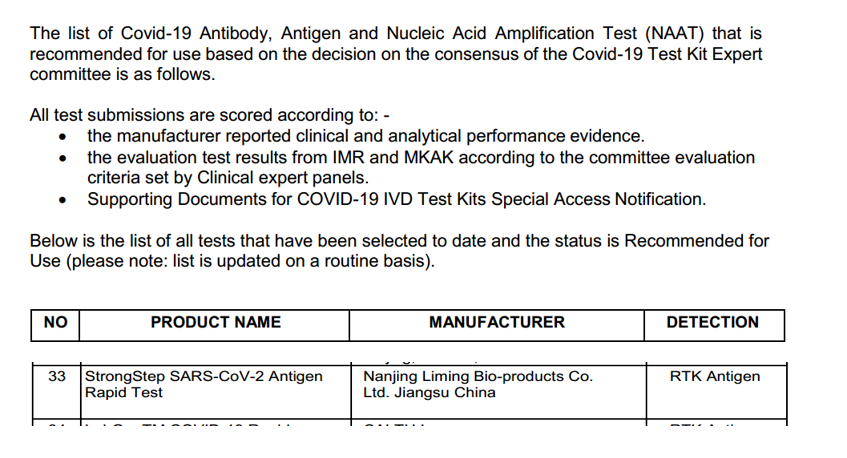
Posachedwa, amphamvu amphamvu ® cov-2 antigen antigen antigen kukula kwa nanzing kukhazikika kwa bio Ntchito zaumunthu (DHSC) yoyesedwa ndikuyamika.
Asanachitike, amphamvu amphamvu Maziko Omwe Analimbikitsa, Germany Federal Agency for Mediction ndi Zipangizo Zachipatala , Chitsimikizo cha Argentina, chiphaso cha Dominica, chitsimikizo cha Guatemala ndi zina. Pakadali pano, South Africa, Illia, yemwe Eul, Eua, FDA, Whitelist wa Fritonist ndi mapulogalamu ena a Chitsimikizo chikuchitika.
Kuyambira Chithunzi: Kulimbikitsidwa ndi Unduna wa Zaumoyo wa Malaysia

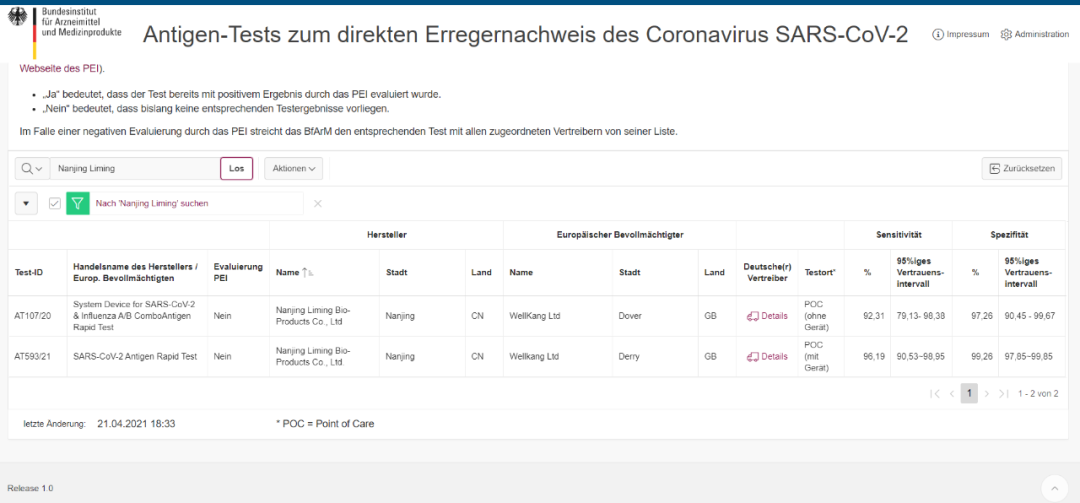
Chithunzi: Singapore HSA Certification

(Gwero la chithunzi: tsamba lovomerezeka la dipatimenti ya Britain yaumoyo ndi ntchito za anthu)
Mu 2020, dipatimenti yaumoyo ndi anthu ogwirizana ku United Kingdom adzatsimikizira motsimikiza madokotala a covid a Covid-19 akulowa m'dziko lokwanira. Pali zinthu 120 kutenga nawo mbali pazotsimikizira, zomwe pazinthu 19 zokha zapereka chitsimikiziro. Pambuyo pa miyezi isanu ndi iwiri yotsimikizika mobwerezabwereza, zonena zowoneka bwino, 200 zonena zabwino ndi zosankha zopitilira 1,000 zimatsimikizira kukula kwa ma nanzing kutsatsa bio-proses.
.
Chilolezo chovomerezeka: AT593 / 21
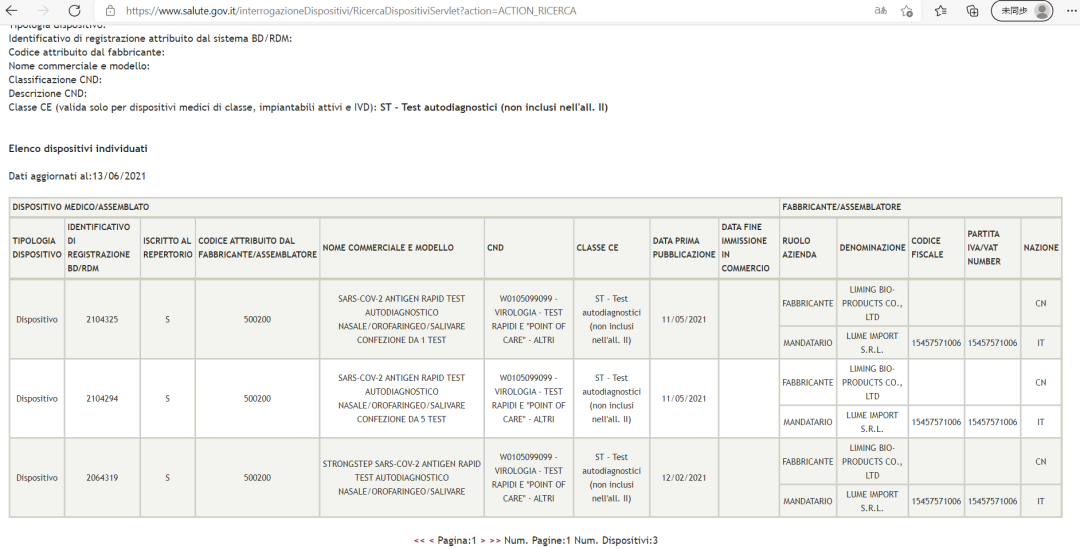
Amphamvu amphamvu® SAV-COV-2 Antigen Final Fir-Sturfied Final (Gulu Lodziyesa) lavomerezedwa ndi Unduna wa Zaumoyo wa Italian
Gwero: Tsamba lovomerezeka la Unduna wa Thanzi Italy laumoyo (Utumiki Della)

Amphamvu amphamvu ®
Kuyesedwa kwa Anzake ndi 2 Ndioyenera kuwunika mwachangu kwa milandu yayikulu ya kachilombo ka HIV yatsopano, makamaka pakuzindikira mwachangu kwa kufalikira kwamphamvu. Itha kugwiritsidwa ntchito ngati mzere woyamba chitetezo, amagwiritsa ntchito matenda oyamba ndi matenda, kuthandiza kupewa kufalikira, ndikuwongolera kufalikira kwa kachilomboka.
Covid-19 lidzakhala m'thupi lokhala mliri mtsogolo, ndipo kufunikira kwa mayeso kudzakula. Zolemba zosiyanasiyana zofunsira, nanjong limadumphira bio-malonda Com., Ltd. adapanga mitundu yosiyanasiyana ya SAR-2, 2 ozindikira antibody + kapena 2 / A ndi B Antigen Phunziro mwachangu + Zowoneka ndi kupewa onse pamsika wapadziko lonse lapansi. Mokwanira amathandizira kupewa kupewa komanso kuwongolera kwa mliri wapadziko lonse lapansi komanso kupewa komanso kuwongolera matenda opuma monga chimfine.
Post Nthawi: Jun-23-2021







